
Non-Invasive Insight
Surface EMG of the diaphragm offers direct, non-invasive access to neural respiratory drive. This is ideal for dynamic patient monitoring.

Macawi SERA is a non-invasive sensing platform that brings real-time visibility into a patient’s respiratory and cardiac activity. By leveraging surface electromyography (sEMG), SERA captures the electrical signals of the diaphragm and heart, enabling clinicians to monitor breathing effort and spontaneous respiratory activity without invasive procedures. This technology supports more responsive and individualized ventilation strategies, helping care teams make timely, informed decisions in critical care environments.
sEMG is a well-established method for assessing muscle activity. Its application in respiratory care enables more personalized and responsive treatment strategies. Other technologies have already demonstrated the clinical relevance of muscle activity-based ventilation. With advances in sensing and signal processing, SERA now makes these insights accessible without invasive procedures.
By visualizing the phases of the breathing cycle in near-real time, SERA supports clinicians in tailoring ventilation strategies to individual patient needs. When combined with cardiac signal monitoring, it provides a broader view of the cardiorespiratory system, contributing to more informed decision-making in critical care environments.

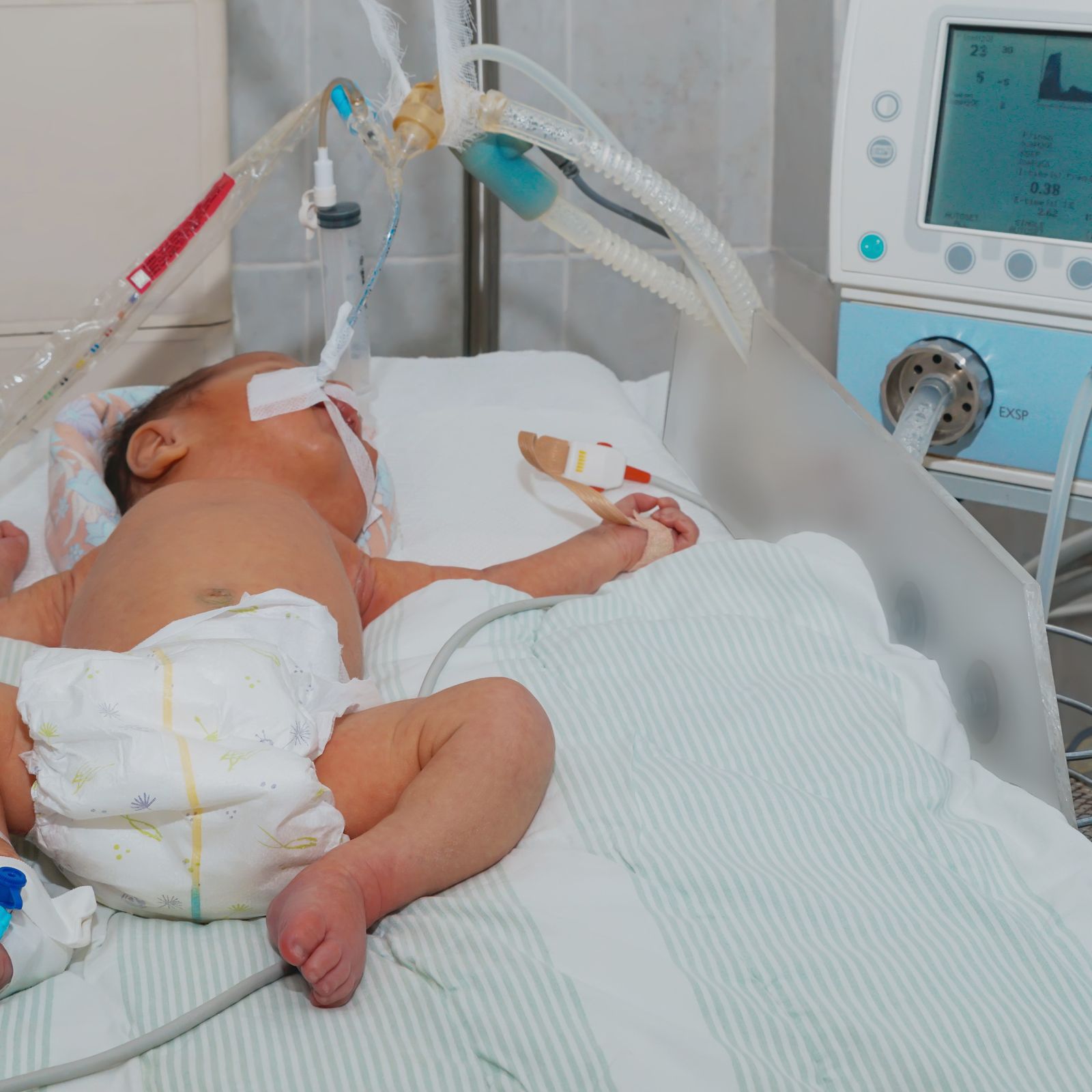
SERA is designed as a stand-alone monitoring system that integrates seamlessly into clinical workflows. All without invasive procedures. Its specially developed electrodes are suitable for all patient populations, from premature infants to adults, and its plug-and-play design ensures compatibility with any ventilator system.
As respiratory care moves toward more personalized and responsive support, SERA stands ready to unlock the full potential of sEMG technology for clinicians, ventilator manufacturers, and healthcare providers worldwide.

Surface EMG of the diaphragm offers direct, non-invasive access to neural respiratory drive. This is ideal for dynamic patient monitoring.

Real-time monitoring of neural respiratory drive enhances understanding of patient condition and therapy response.
Dual-function sEMG monitoring of diaphragm and heart—SERA enables precise, non-invasive cardiorespiratory insights.

Intuitive setup with familiar ECG-like design, just one extra lead for powerful dual-function monitoring.
The Macawi SERA sensor can be used in all patient groups. Ranging from the smallest preterms to the largest adults in ICU-environments.
"Macawi SERA transforms respiratory care by making real-time, personalized monitoring accessible to every clinician. No invasive procedures, no guesswork. Let’s explore how this technology can support your team and your patients"
